Inspired by Lui et al 2021.[1]
EA – electroacupuncture
key to acronyms
IF – impact factor (2-year)
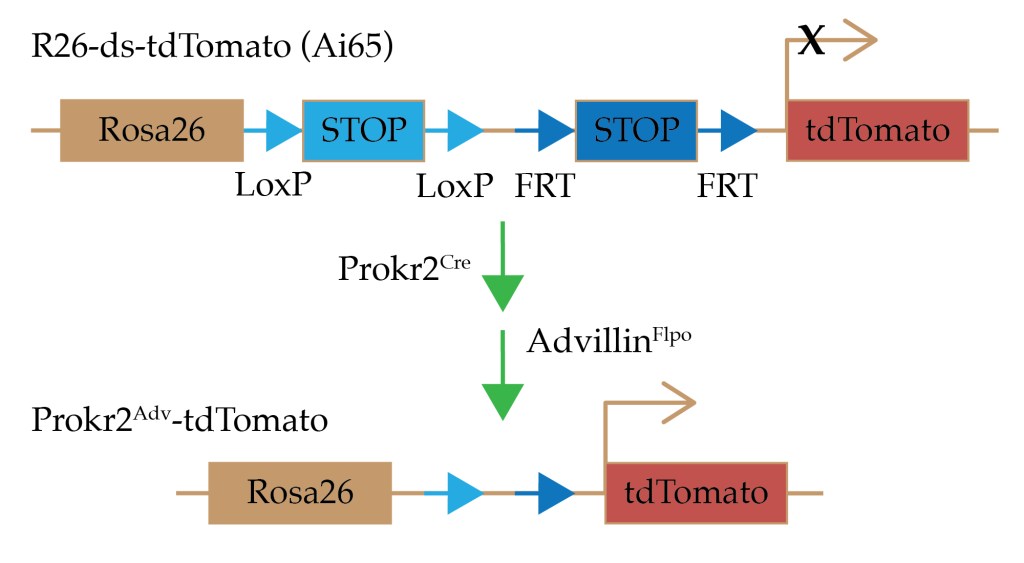
Prokr2Adv – a set of nerves expressing both Prokr2 and advillin
Prokr2 – prokineticin receptor 2 (a G protein-coupled receptor)
Advillin – a sensory neuron-specific protein functionally linked to β-actin
TNF – tumour necrosis factor (a proinflammatory cytokine)
IL-6 – interleukin 6 (a proinflammatory cytokine)
LLLT – low level laser therapy
CatCh – calcium-translocating channel rhodopsin
rhodopsin – a light sensitive receptor protein
eYFP – enhanced yellow fluorescent protein emitting at 527nm
tdTomato – a fluorescent protein emitting at 581nm (so it looks tomato coloured)
Oh my word, what a fabulous surprise to be sent this paper (by Ari Moré) before it showed up on my daily searches. It is a full article on EA in the journal Nature (IF 49.962).
Nature – 2 year IF is 49.962
2020 journal metrics
Nature – 5 year IF is 54.637
We have never seen such a paper in this highly prestigious journal. In fact we haven’t seen any paper in this journal with either acupuncture or electroacupuncture in the title for over 40 years,[2–4] apart from a handful of news reports.[5–7]
This paper follows on from the rather complex one in Neuron,[8] which I commented on here in September 2020: ST36 vs ST25 EA in sepsis. Thankfully I remember some the genetic manipulation techniques, so this paper was easier to read.
Essentially the team used the protective effect of EA in a mouse model of sepsis that we were first introduced to by Luis Ulloa and colleagues in 2014.[9] In the Neuron paper, Qiufu Ma and his team identified a subset of sympathetic nerves that mediated both increased and reduced lethality in the model depending on intensity and timing of stimulation at ST25. In this paper they have identified a nerve cell line responsible for the protective effect of low intensity (0.5mA) EA (Prokr2Adv), which only works in certain somatic regions including deep tibialis anterior (ST36) and deep in the wrist extensors (LI10), but not in others, such as lateral gastrocnemius and semitendinosus or superficial tissues or near cutaneous nerves.
This will inevitably lead to cries of point specificity from acupuncture advocates, but we need to be careful how we use this data, since the areas innervated by the identified nerves are distributed widely through deep connective tissue in the limbs, and not concentrated at the points studied.
the idea of point specificity
I first came across a renowned physiologist insisting on advocating the idea of point specificity when editing a chapter for a multi author textbook.[10] The late John Longhurst’s investigations of cardiovascular reflexes in cats demonstrated that EA could only obtund the excitatory reflexes when applied at some acupuncture points but not at others.[11] The difference was the points in deep somatic tissue worked, but those that stimulated sensory nerves to skin did not.
This paper in Nature takes the concept of regional tissue specificity further by identifying superficial muscles as well as other superficial structures that are not able to mediate anti-inflammatory effects at low levels of stimulation. At high levels of stimulation (3.0mA) there was no such distinction, and the effect was not mediated by the specific nerves identified (Prokr2Adv).
Another feature of this paper that surprised me was the use of laser. I have long had an interest in laser, but always considered it to be quite different from needle acupuncture or EA in physiological terms. They used laser applied inside the tibialis anterior muscle at ST36 to achieve the same effect as the low intensity EA in terms of anti-inflammatory effects (reduced serum TNF and IL6) and improved survival in the mouse sepsis model.
Now, before the laser advocates among you try to treat sepsis with LLLT at ST36, this only worked in a specifically bred line of mice in which the nerves initiating the anti-inflammatory effects (Prokr2Adv) were labelled with special calcium translocating channels (CatCH) containing rhodopsin plus a yellow fluorescent protein (eYFP) to identify them. Also note that the lasers were buried deep into the muscle, which rather negates the obvious advantages of using LLLT in needle phobic patients.
tdTomato – a fluorescent protein
emitting photons with a wavelength of 581nm
Now that I have mentioned eYFP, I cannot finish this blog without mentioning tdTomato. It caused me a lot of trouble when I first came across it in the Neuron paper, principally because it does not have its own page on Wikipedia. You will find it on the page titled: Fluorophore, but it is still hard to find on that page since it is one of a great long list of other fluorescent proteins.
The process by which it is specifically expressed and therefore lights up the cell for histological identification is a bit complicated, but I will do my best to simplify the genetics at the webinar on Wednesday. Don’t miss it or you’ll have to spend ages trying to get your head around Cre–Lox recombination and flippases without my help ;-).
Importantly, the same process is used to activate a receptor for diphtheria toxin in a highly specific group of nerve cells (Prokr2Adv), to allow selective ablation and demonstrate that the effect of low intensity EA is abolished.
References
1 Liu S, Wang Z, Su Y, et al. A neuroanatomical basis for electroacupuncture to drive the vagal-adrenal axis. Nature Published Online First: 13 October 2021. doi:10.1038/s41586-021-04001-4
2 Cheng SB, Ding LK. Practical application of acupuncture analgesia. Nature 1973;242:559–60. doi:10.1038/242559a0
3 Peets JM, Pomeranz B. CXBK mice deficient in opiate receptors show poor electroacupuncture analgesia. Nature 1978;273:675–6. doi:10.1038/273675a0
4 Chung SH, Dickenson A. Pain, enkephalin and acupuncture. Nature 1980;283:243–4. doi:10.1038/283243a0
5 Acupuncture boom punctured. Nature 1985;318:222.
6 Cressey D. Acupuncture for mice. Nature 2010;465:538. doi:10.1038/465538a
7 Marchant J. Acupuncture in cancer study reignites debate about controversial technique. Nature 2017;552:157–8. doi:10.1038/d41586-017-08309-y
8 Liu S, Wang Z-F, Su Y-S, et al. Somatotopic Organization and Intensity Dependence in Driving Distinct NPY-Expressing Sympathetic Pathways by Electroacupuncture. Neuron 2020;108:436-450.e7. doi:10.1016/j.neuron.2020.07.015
9 Torres-Rosas R, Yehia G, Peña G, et al. Dopamine mediates vagal modulation of the immune system by electroacupuncture. Nat Med 2014;20:291–5. doi:10.1038/nm.3479
10 Longhurst JC. Acupuncture in cardiovascular medicine. In: Filshie J, White A, Cummings M, eds. Medical Acupuncture – A Western Scientific Approach. 2nd edition. London: Elsevier, 2016. p. 394–421.
11 Tjen-A-Looi SC, Fu L-W, Guo Z-L, et al. Modulation of Neurally Mediated Vasodepression and Bradycardia by Electroacupuncture through Opioids in Nucleus Tractus Solitarius. Sci Rep 2018;8:1900. doi:10.1038/s41598-018-19672-9
You must be logged in to post a comment.