Inspired by Friedemann et al 2022.[1]
CIPN – chemotherapy-induced peripheral neuropathy
key to acronyms
NCS – nerve conduction studies
MA – manual acupuncture
EA – electroacupuncture
SNAP – sensory nerve action potential
SNCV – sensory nerve conduction velocity
PROMs – patient reported outcome measures
MYMOP2 – measure yourself medical outcome profile 2
I think I heard one of the authors of this study present the results at a meeting towards the beginning of 2022. It is a relatively small study (n=60) but it is the first to use NCS as the primary outcome in a trial of acupuncture for CIPN.
I have a blog category for CIPN already, so this will add a 5th post.
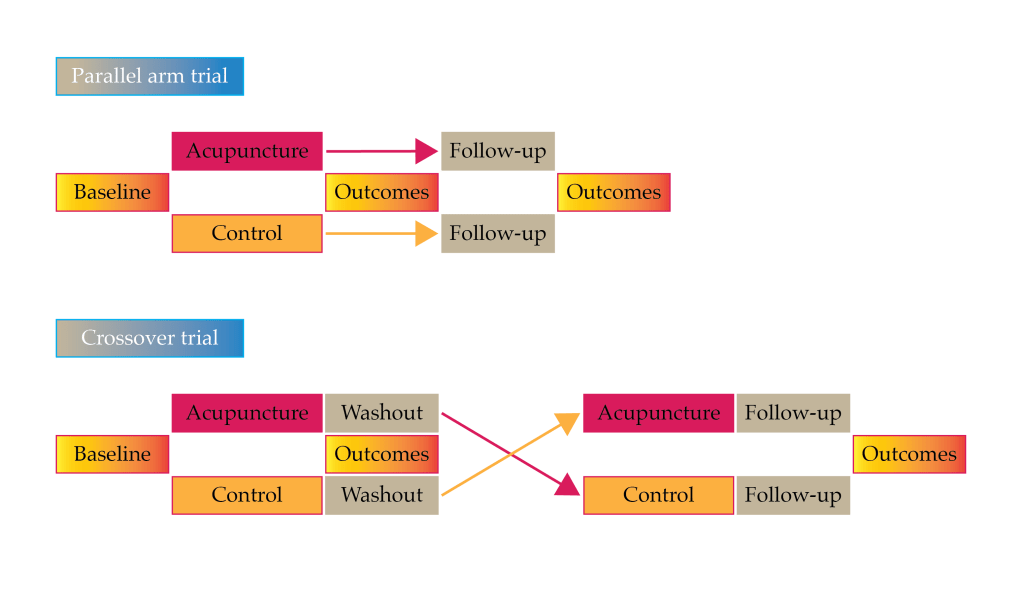
It is unusual in that it uses a crossover design. Crossover designs have a big advantage in terms of statistical power but a big disadvantage if your treatment has a long duration of effect. In acupuncture studies this can be a problem, although it is a desirable one for most patients. It will all depend on the outcome measure used and the length of the washout period. In this case it looks as though the authors only used change figures, so an improvement that was maintained from the active phase into the control phase may not disadvantage the active treatment. I don’t know why I have never thought of this before as a solution to the crossover trial dilemma in acupuncture!
Crossover design:
advantage – statistical power
disadvantage – inadequate washout period
Keen-eyed readers will have already spotted that this paper was published in the journal Phytomedicine, which seems a little odd for an acupuncture paper. The impact factor of Phytomedicine is 6.656, which is somewhat higher than all acupuncture journals and the majority in the field of complementary medicine. A couple of the authors had already published papers in this journal, so I guess the editors allowed a little flexibility in the published journal scope in this case.
Phytomedicine
Impact Factor 6.656
Talking of acupuncture journals, one of the team had already published an observation (n=6) regarding the effects of MA on CIPN in Acupuncture in Medicine.[2] This paper described improvements in NCS in 5 out of the 6 patients. This author also collaborated in a 4-arm trial of the effectiveness of EA in CIPN (n=60), although this study failed to show any differences between the 4 groups, one of which was placebo capsules.[3]
In the current trial, the NCS at baseline were more abnormal than in the neutral 4-arm trial,[3] but not as abnormal as those in the cohort of 6,[2] most of which had unrecordable values for sural SNAP and SNCV.
The points used were the same as in the cohort from 2012 – ST34, Bafeng, Qiduan. Baxie and Shixuan were added if the upper limbs were also affected. I use Baxie and Bafeng points quite a lot in CIPN. These are the extra points between the metacarpal and metatarsal heads respectively – there are 4 in each extremity. I do not use the extra points Shixuan and Qiduan, which are in the very ends of the fingers and toes respectively.
MA was applied once a week for 10 weeks followed by a 4-week washout period. The acupuncture group then swapped over to 10 weeks of control (no treatment), and the control group received 10 sessions of acupuncture over a further 10 weeks. NCS were performed before randomisation and then again after each phase of the crossover trial – 3 times in total.
In terms of the NCS, the only abnormalities at baseline were in measurements from the sural nerve – SNAP amplitude and SNCV (note that the SN in the acronyms stands for sensory nerve rather than sural nerve). Motor nerve measurements of the tibial nerve were all in the normal range.
During the acupuncture phases of the trial the mean sural SNAP (primary outcome) almost doubled in amplitude, and the mean SNCV increased by around 25%, which put it well into the normal range since at baseline the mean was only marginally in the abnormal range.
SNAP amplitude almost doubled…
It is interesting to note that whilst all PROMs (symptom scores) improved with MA, only 5 of the 12 measures reached significance. This emphasises the importance of being very careful in choosing the primary outcome for such a trial. Similar, only 4 of 9 different measures in clinical neurological signs improved significantly after active treatment (MA).
This trial was labelled with the acronym ACUCIN, which I ignored at first. It was only when I was checking other publications by the same authors that I happened upon ACUDIN. The latter acronym refers to a larger (n=180) 3-arm trial of acupuncture in diabetic peripheral neuropathy published in 2021 – another paper I clearly overlooked.[4]
The 3 arms in this study were MA (same points listed above), laser acupuncture and placebo laser acupuncture. This was a parallel 3 arm study with 60 in each arm, so in terms of comparisons between individual groups the statistical power is rather similar to the crossover trial with 30 in each group. Change in sural SNAP was again the primary outcome, and the trial demonstrated a significant effect for both MA and laser over placebo. MA showed a trend to be a better than laser in most outcomes with some reaching significance.
SNAP amplitude improvement probably indicates structural neural regeneration
As the authors comment in the conclusions of both trials, improvement in sural SNAP amplitude is consistent with structural neural regeneration. This is important because we do not have disease modifying therapies for these peripheral neuropathies… until now perhaps.
Addendum
A day or so after finishing this blog post, I was informed about the publication of another trial in CIPN. This time a pragmatic trial (n=120) from the Christie Hospital in Manchester, UK.[5] A 2 point or greater improvement in MYMOP2 score was used to define response to treatment. The group randomised to receive ten sessions of acupuncture in addition to usual care had a responder rate of 68% compared with 33% in the group randomised to usual care alone.
References
1 Friedemann T, Kark E, Cao N, et al. Acupuncture improves chemotherapy-induced neuropathy explored by neurophysiological and clinical outcomes – The randomized, controlled, cross-over ACUCIN trial. Phytomedicine 2022;104:154294. doi:10.1016/j.phymed.2022.154294
2 Schroeder S, Meyer-Hamme G, Epplée S. Acupuncture for chemotherapy-induced peripheral neuropathy (CIPN): a pilot study using neurography. Acupunct Med 2012;30:4–7. doi:10.1136/acupmed-2011-010034
3 Rostock M, Jaroslawski K, Guethlin C, et al. Chemotherapy-induced peripheral neuropathy in cancer patients: a four-arm randomized trial on the effectiveness of electroacupuncture. Evid-Based Complement Altern Med ECAM 2013;2013:349653. doi:10.1155/2013/349653
4 Meyer-Hamme G, Friedemann T, Greten J, et al. Electrophysiologically verified effects of acupuncture on diabetic peripheral neuropathy in type 2 diabetes: The randomized, partially double-blinded, controlled ACUDIN trial. J Diabetes 2021;13:469–81. doi:10.1111/1753-0407.13130
5 Stringer J, Ryder WD, Mackereth PA, et al. A randomised, pragmatic clinical trial of ACUpuncture plus standard care versus standard care alone FOr Chemotherapy Induced peripheral Neuropathy (ACUFOCIN). Eur J Oncol Nurs 2022;60:102171. doi:10.1016/j.ejon.2022.102171
You must be logged in to post a comment.