Inspired by Ball et al 2022.[1]
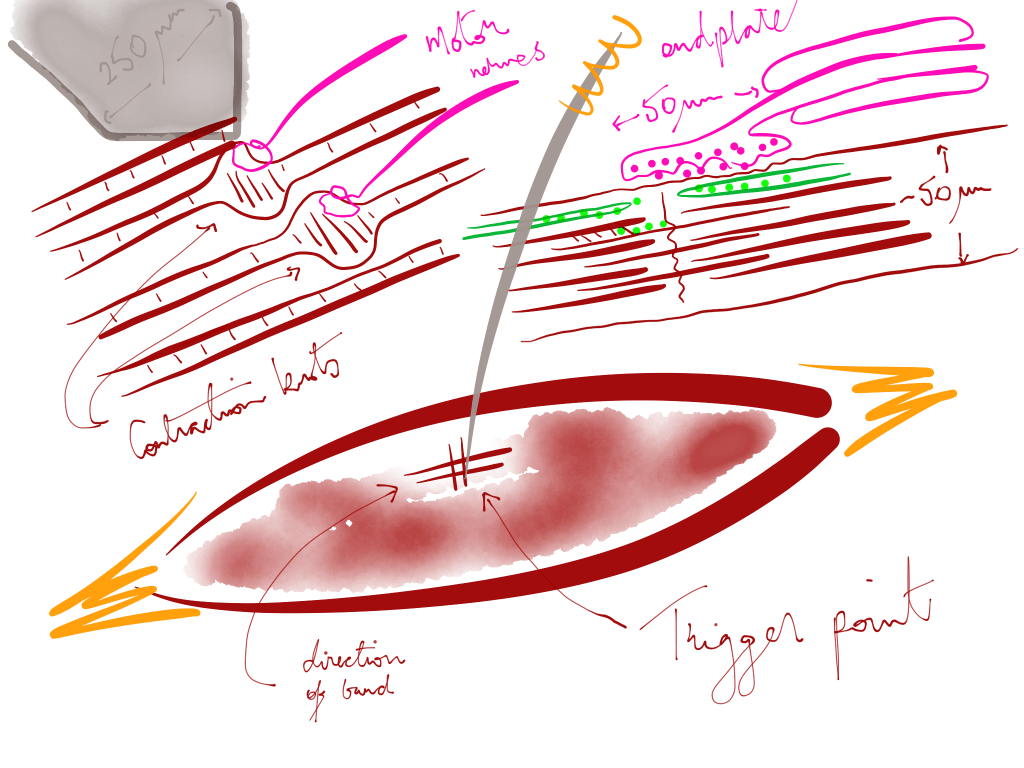
MTrP – myofascial trigger point
key to acronyms
TrP – trigger point (exactly the same as MTrP for those wanting to save a keystroke)
LTR – local twitch response
US – ultrasound
VSE – vibration sonoelastography
SWE – shear wave elastography
DN – dry needling aka trigger point needling
CEUS – contrast enhanced ultrasound
DCS – decompression sickness
Trigger points, either MTrPs or TrPs, were my first main interest when I swapped my fine hypodermic needles for the even finer solid filiform ones (ie acupuncture needles). Following on from my much-increased use of needling was an interest in the referred pain patterns produced from needling different somatic structures.
Referred pain produced by needling in a primary TrP could be diagnostic if it was clearly recognised by the patient and if the spontaneous pain resolved after needling. Eliciting an LTR at the same time seemed to be associated with a positive outcome in my hands, but this is still somewhat controversial.[2]
This paper describes and interprets for the first time the detailed architecture seen with high-definition US imaging of TrPs from two different patient cases. The findings suggest a reason why repeated needling in the same area produces multiple LTRs in some patients, and that this may be the best approach for the less sensitive individuals with recalcitrant myofascial pain.
The first attempt to reliably detect TrPs with US was published in the late 90s in the journal Pain and was unsuccessful.[3] But just under a decade later a group led by Lynn Gerber in Fairfax, Virginia came up with the idea of applying vibration at the same time as imaging and the stiffer area of the TrP lit up like a black hole.[4] I apologise for the incongruent metaphors. You will understand when you see the typical US images. Generally, muscle looks like a lot of white lines on US, but the TrP shows up as a hypoechoic region (a black hole in the image).
the TrP lit up like a black hole
This group went on to study the differences between active TrPs, latent ones and normal muscle in a blinded evaluation using VSE with colour Doppler.[5] This led to further interest in the blood flow in and around the TrP.[6]
A little later it was a group in Dublin that demonstrated an immediate reduction in stiffness of TrPs using SWE following a single session of needling (DN) performed in the upper trapezius of 7 women.[7] This was repeated by the group from Fairfax in a more comprehensive study on 48 people with chronic myofascial pain.[8] This study utilised 3 sessions of DN and made measurements of stiffness at baseline, 3 weeks and 8 weeks.
Having failed initially in the late 90s,[3] some two decades later with fancier kit we can now reliably find TrPs using US,[9] and can even differentiate active from latent TrPs with something called texture analysis.[10]
Finally, a group in Beijing has even used CEUS in addition to SWE to identify TrPs for biopsy.[11] CEUS uses an intravascular injection of a solution containing lots of tiny bubbles at the same time as performing US and is used as a way of visualising vascular blood volume and transit time.
I have a vague memory of doing something similar to look for significant right to left atrial shunts in commercial divers following an episode of the bends (DCS). I would withdraw ~9mls of blood add ~1ml of air and shake the syringe until there was a homogeneous dark red foamy appearance. When the sonographer had a stable image of the chambers of the heart, I would inject the foamy blood back through the cannula and a few seconds later we would see the shower of bubbles whizz through the right side of the heart. I confess it was slightly counter intuitive to inject air bubbles into a vein, but I trust they all got mopped up in the lungs!
lots of tiny hyperechoic speckles
So, getting back to the latest paper, this has demonstrated that the typical hypoechoic appearance of the TrP is actually filled by lots of tiny hyperechoic speckles. The authors propose that this is compatible with the multiple loci hypothesis of Hong and Simons.[12]
Hypoechogenicity of the TrP nodule is associated with hyperperfusion, but the hyperechogenicity of the speckles is consistent with ischaemic areas within the nodule.
These are exciting times as the denial of the TrP becomes an ever more tenuous position for a clinician to hold.
References
1 Ball A, Perreault T, Fernández-de-Las-Peñas C, et al. Ultrasound Confirmation of the Multiple Loci Hypothesis of the Myofascial Trigger Point and the Diagnostic Importance of Specificity in the Elicitation of the Local Twitch Response. Diagn Basel Switz 2022;12:321. doi:10.3390/diagnostics12020321
2 Perreault T, Dunning J, Butts R. The local twitch response during trigger point dry needling: Is it necessary for successful outcomes? J Bodyw Mov Ther 2017;21:940–7. doi:10.1016/j.jbmt.2017.03.008
3 Lewis J, Tehan P. A blinded pilot study investigating the use of diagnostic ultrasound for detecting active myofascial trigger points. Pain 1999;79:39–44. doi:10.1016/S0304-3959(98)00155-9
4 Sikdar S, Shah JP, Gilliams E, et al. Assessment of myofascial trigger points (MTrPs): a new application of ultrasound imaging and vibration sonoelastography. Annu Int Conf IEEE Eng Med Biol Soc 2008;2008:5585–8. doi:10.1109/IEMBS.2008.4650480
5 Sikdar S, Shah JP, Gebreab T, et al. Novel applications of ultrasound technology to visualize and characterize myofascial trigger points and surrounding soft tissue. Arch Phys Med Rehabil 2009;90:1829–38. doi:10.1016/j.apmr.2009.04.015
6 Sikdar S, Ortiz R, Gebreab T, et al. Understanding the vascular environment of myofascial trigger points using ultrasonic imaging and computational modeling. Annu Int Conf IEEE Eng Med Biol Soc 2010;2010:5302–5. doi:10.1109/IEMBS.2010.5626326
7 Maher RM, Hayes DM, Shinohara M. Quantification of dry needling and posture effects on myofascial trigger points using ultrasound shear-wave elastography. Arch Phys Med Rehabil 2013;94:2146–50. doi:10.1016/j.apmr.2013.04.021
8 Turo D, Otto P, Hossain M, et al. Novel Use of Ultrasound Elastography to Quantify Muscle Tissue Changes After Dry Needling of Myofascial Trigger Points in Patients With Chronic Myofascial Pain. J Ultrasound Med 2015;34:2149–61. doi:10.7863/ultra.14.08033
9 Kumbhare D, Shaw S, Grosman-Rimon L, et al. Quantitative Ultrasound Assessment of Myofascial Pain Syndrome Affecting the Trapezius: A Reliability Study. J Ultrasound Med 2017;36:2559–68. doi:10.1002/jum.14308
10 Kumbhare DA, Ahmed S, Behr MG, et al. Quantitative Ultrasound Using Texture Analysis of Myofascial Pain Syndrome in the Trapezius. Crit Rev Biomed Eng 2018;46:1–31. doi:10.1615/CritRevBiomedEng.2017024947
11 Liang X-N, Guo R-J, Li S. New application of multimodal ultrasound imaging for identification of myofascial trigger points in the trapezius muscle. Ann Palliat Med 2021;10:9784–91. doi:10.21037/apm-21-2103
12 Hong CZ, Simons DG. Pathophysiologic and electrophysiologic mechanisms of myofascial trigger points. Arch Phys Med Rehabil 1998;79:863–72. doi:10.1016/s0003-9993(98)90371-9
You must be logged in to post a comment.